For over a century, biology has rested on a singular and poetic metaphor: the Tree of Life. We picture a single and primordial trunk, one definitive "common ancestor," from which all complexity eventually branched out. But a revolutionary theory presented by microbiologist Carl R. Woese suggests that if we look back far enough the tree disappears entirely. In its place is not a single organism but a roiling global ecosystem where individual identity did not exist and genes flowed as freely as water.
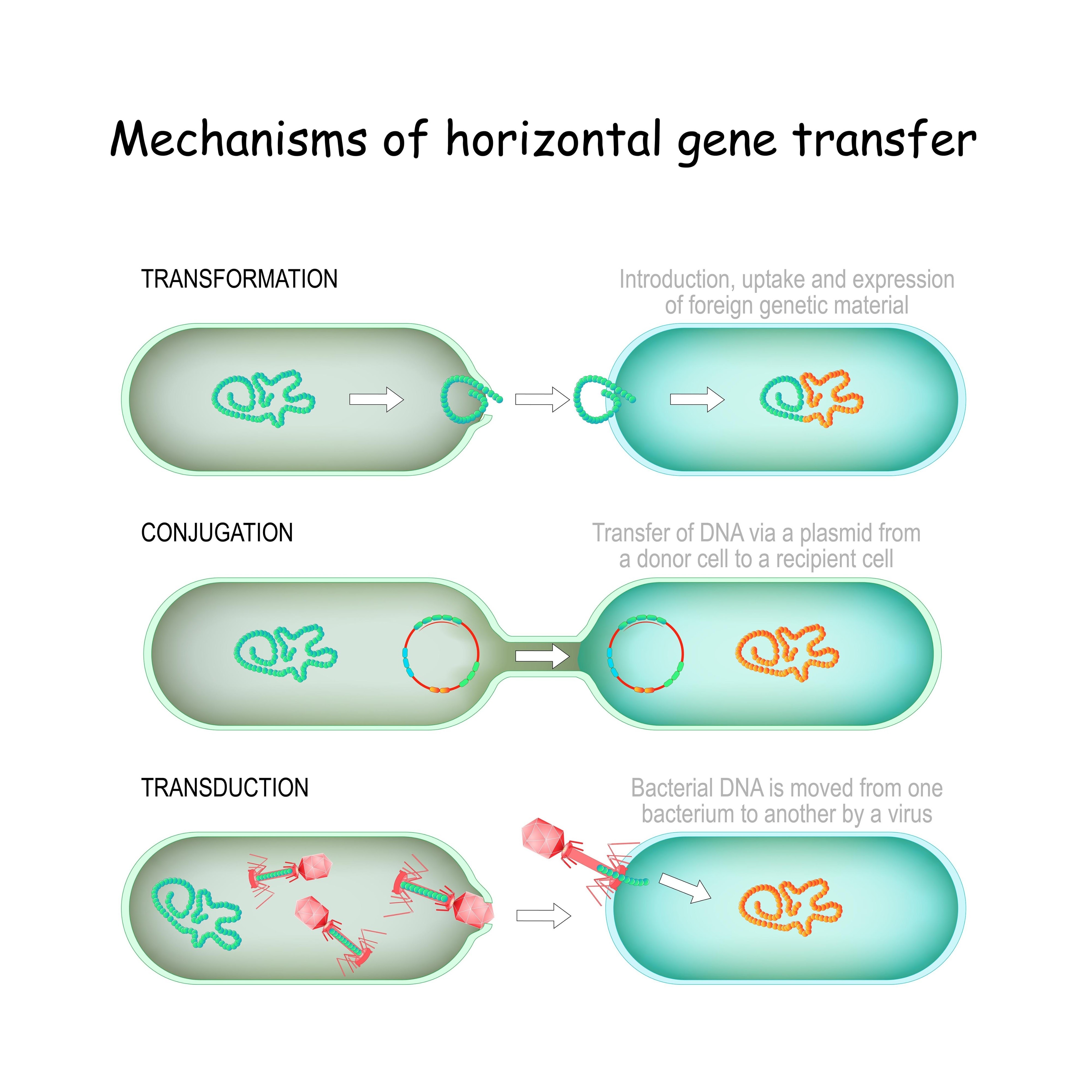
In his paper "On the evolution of cells," Woese argues that the classical Darwinian model, which relies on vertical inheritance from parent to offspring, breaks down when we try to explain the very origin of cells. Instead he proposes that early life was driven by a force that modern biology often overlooks: Horizontal Gene Transfer (HGT).
The Era of Genetic Chaos
To understand the origin of life we have to unlearn the rules of modern biology. Today a cell is a fortress. It has a complex and integrated genome that is difficult to alter without killing the organism. But Woese posits that "aboriginal" cell designs were far simpler and looser. They were modular like structures built from Lego bricks that could be easily snapped apart and reassembled.
In this primitive "RNA world" cells were not distinct species with permanent genetic histories. They were ephemeral vessels in a "universal gene-exchange pool". If a neighbor developed a useful protein or a better way to replicate, that innovation was not locked away in a private lineage. It was swapped, stolen, and shared horizontally across the entire community. Evolution was not about the survival of the fittest individual lineage. It was a matter of "communal invention" where the entire ecosystem evolved as a single and massive entity.
Shutterstock
gene transfer. horizontal Mechanisms
Crossing the Darwinian Threshold
This chaotic and communal state could not last forever. As cells scavenged the best machinery from their neighbors they inevitably became more complex. Their internal systems, like translation, transcription, and replication, became tightly coupled and interdependent. A cell could no longer swap out a core component for a foreign one without causing the whole system to crash.
Woese calls this critical turning point the "Darwinian Threshold". It represents a phase change in the organization of life. Once a cell crossed this threshold its genetic history solidified. It stopped being a permeable vessel and became a distinct lineage capable of "speciation" as we know it today.
This theory radically reinterprets the "root" of the universal phylogenetic tree. The root is not a single and specific organism that lived at a specific time. Instead the root represents the Darwinian Threshold itself, the moment when the universal gene pool crystallized into distinct streams.
Three Paths Out of the Pool
Perhaps the most startling implication of Woese’s model is that the three primary domains of life—Bacteria, Archaea, and Eucarya—did not branch off from one another in a simple sequence. Instead they likely emerged independently from that ancestral communal pool.
The Bacteria were likely the first to cross the threshold and "solidify" their cellular design which is why their machinery looks the most primitive. The Archaea and Eucarya remained in the communal pool longer. They continued to swap and refine their information processing systems before eventually crossing their own thresholds and becoming distinct domains. This explains why, despite their differences, these groups share eerily similar genetic machinery. They were forged in the same "high evolutionary temperatures" of the ancestral community.
Woese’s perspective forces us to abandon the "Doctrine of Common Descent" in its strictest sense. Life on Earth does not descend from one primordial form but from a process. We are the children of a global community that innovated together, swapped secrets, and eventually grew apart to build the biological world we know today.
Primary Reference:Woese, C. R. (2002). "On the evolution of cells." Proceedings of the National Academy of Sciences, 99(13), 8742-8747.